上海金畔生物科技有限公司为生命科学和医药研发人员提供生物活性分子抑制剂、激动剂、特异性抑制剂、化合物库、重组蛋白,专注于信号通路和疾病研究领域。
AKN-028
AKN-028 是具有口服活性的酪氨酸激酶的抑制剂 (TKI),是 FLT3 的抑制剂,其 IC50 值为 6 nM。AKN-028 可剂量依赖性的诱导 FLT3 的自磷酸化。
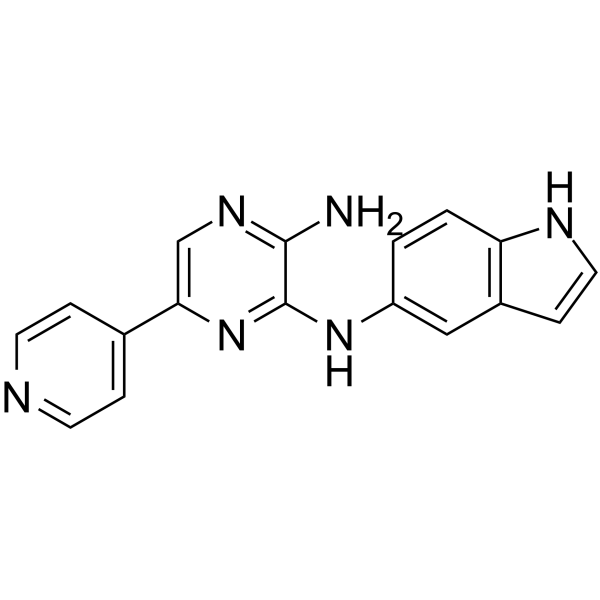
AKN-028 Chemical Structure
CAS No. : 1175017-90-9
| 规格 | 是否有货 | ||
|---|---|---|---|
| 100 mg | 询价 | ||
| 250 mg | 询价 | ||
| 500 mg | 询价 |
* Please select Quantity before adding items.
| 生物活性 |
AKN-028 is an orally active and potent FLT3 tyrosine kinase inhibitor (IC50 = 6 nM). AKN-028 causes dose-dependent inhibition of FLT3 autophosphorylation[1]. |
||||||||
|---|---|---|---|---|---|---|---|---|---|
| IC50 & Target |
|
||||||||
| 体外研究 (In Vitro) |
AKN-028 triggers apoptosis in MV4-11 by activation of caspase 3[1]. 上海金畔生物科技有限公司 has not independently confirmed the accuracy of these methods. They are for reference only. Cell Cytotoxicity Assay[1]
|
||||||||
| 体内研究 (In Vivo) |
AKN-028 (15 mg/kg, Subcutaneously injection twice daily) exhibits anti-tumor activities for acute myeloid leukemia (AML) models[1]. 上海金畔生物科技有限公司 has not independently confirmed the accuracy of these methods. They are for reference only.
|
||||||||
| Clinical Trial |
|
||||||||
| 分子量 |
302.33 |
||||||||
| Formula |
C17H14N6 |
||||||||
| CAS 号 |
1175017-90-9 |
||||||||
| 运输条件 |
Room temperature in continental US; may vary elsewhere. |
||||||||
| 储存方式 |
Please store the product under the recommended conditions in the Certificate of Analysis. |
||||||||
| 参考文献 |
|
所有产品仅用作科学研究或药证申报,我们不为任何个人用途提供产品和服务
