上海金畔生物科技有限公司为生命科学和医药研发人员提供生物活性分子抑制剂、激动剂、特异性抑制剂、化合物库、重组蛋白,专注于信号通路和疾病研究领域。
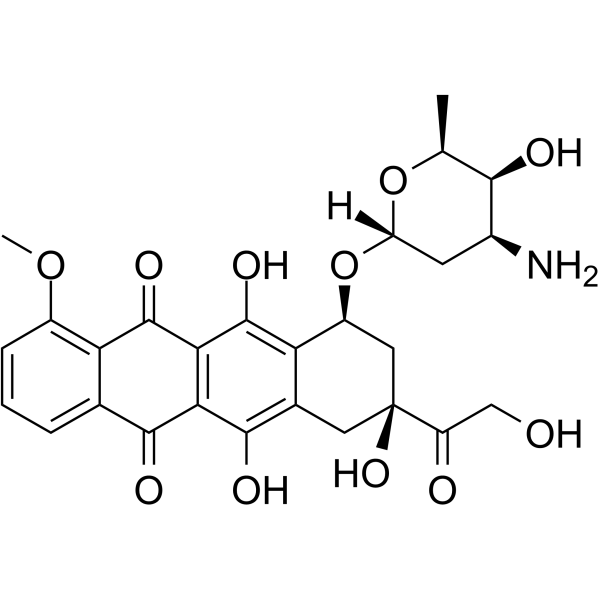
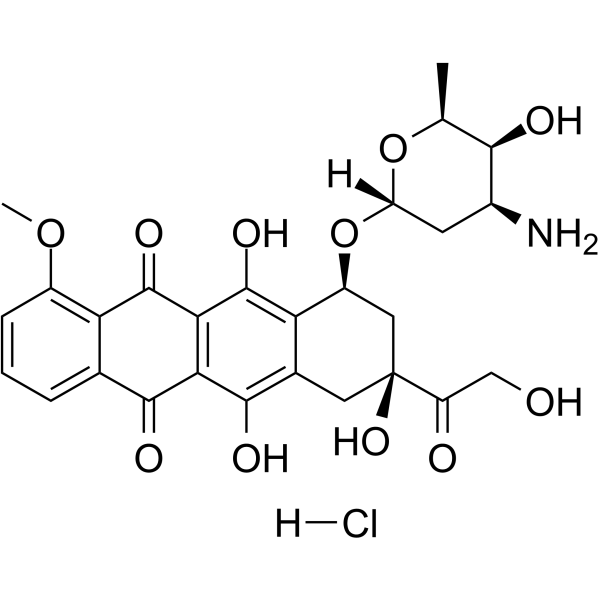
Doxorubicin hydrochloride (Synonyms: 盐酸阿霉素; Hydroxydaunorubicin hydrochloride) 纯度: 98.44%
Doxorubicin (Hydroxydaunorubicin) hydrochloride是一种具有细胞毒性的蒽环类抗生素,是一种抗癌化疗试剂。Doxorubicin hydrochloride 是一种有效的人类 DNA topoisomerase I 和 topoisomerase II 抑制剂,IC50 分别为 0.8 μM 和 2.67 μM。Doxorubicin hydrochloride 可降低 AMPK 及其下游靶蛋白乙酰辅酶 A 羧化酶的磷酸化。还可诱导凋亡 (apoptosis) 和自噬。
Doxorubicin hydrochloride Chemical Structure
CAS No. : 25316-40-9
| 规格 | 价格 | 是否有货 | 数量 |
|---|---|---|---|
| Free Sample (0.1-0.5 mg) | Apply now | ||
| 10 mM * 1 mL in DMSO | ¥990 | In-stock | |
| 50 mg | ¥900 | In-stock | |
| 100 mg | ¥1600 | In-stock | |
| 200 mg | ¥3000 | In-stock | |
| 500 mg | ¥5000 | In-stock | |
| 1 g | ¥8500 | In-stock | |
| 5 g | 询价 | ||
| 10 g | 询价 |
* Please select Quantity before adding items.
Doxorubicin hydrochloride 相关产品
•相关化合物库:
- Covalent Screening Library Plus
- Natural Product Library Plus
- Drug Repurposing Compound Library Plus
- FDA-Approved Drug Library Plus
- FDA-Approved Drug Library Mini
- Bioactive Compound Library Plus
- Anti-Infection Compound Library
- Apoptosis Compound Library
- Cell Cycle/DNA Damage Compound Library
- Epigenetics Compound Library
- Kinase Inhibitor Library
- PI3K/Akt/mTOR Compound Library
- Natural Product Library
- FDA-Approved Drug Library
- Toxins for Antibody-Drug Conjugate Research Library
- Anti-Cancer Compound Library
- Antiviral Compound Library
- Autophagy Compound Library
- Anti-Aging Compound Library
- Drug Repurposing Compound Library
- Covalent Screening Library
- Antioxidants Compound Library
- Oxygen Sensing Compound Library
- Antibacterial Compound Library
- NMPA-Approved Drug Library
- Glycolysis Compound Library
- Cytoskeleton Compound Library
- FDA Approved & Pharmacopeial Drug Library
- Antibiotics Library
- Anti-Lung Cancer Compound Library
- Drug-Induced Liver Injury (DILI) Compound Library
- Anti-Pancreatic Cancer Compound Library
- Anti-Blood Cancer Compound Library
- Anti-Cancer Metabolism Compound Library
- Microbial Metabolite Library
- Mitochondria-Targeted Compound Library
- Lipid Metabolism Compound Library
- Glucose Metabolism Compound Library
- Animal Disease Model Inducer Library
- Tumorigenesis Related Compound Library
- Rare Diseases Drug Library
- Children’s Drug Library
| 生物活性 |
Doxorubicin (Hydroxydaunorubicin) hydrochloride, a cytotoxic anthracycline antibiotic, is an anti-cancer chemotherapy agent. Doxorubicin hydrochloride is a potent human DNA topoisomerase I and topoisomerase II inhibitor with IC50s of 0.8 μM and 2.67 μM, respectively. Doxorubicin hydrochloride reduces basal phosphorylation of AMPK and its downstream target acetyl-CoA carboxylase. Doxorubicin hydrochloride induces apoptosis and autophagy[1][2][3]. |
||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| IC50 & Target[1][2][3] |
|
||||||||||||||||||||||||
| 体外研究 (In Vitro) |
Doxorubicin hydrochloride (1-8 µM; 24 and 48 hours) decreases the viability of MCF-10F, MCF-7 and MDA-MB-231 cells in a time- and dose-dependent manner[4]. 上海金畔生物科技有限公司 has not independently confirmed the accuracy of these methods. They are for reference only. Cell Viability Assay[4]
Cell Cycle Analysis[5]
Western Blot Analysis[4]
|
||||||||||||||||||||||||
| 体内研究 (In Vivo) |
Treatment with Doxorubicin (2 mg/kg) or Zoledronic acid (100 μg/kg) alone does not statistically significantly decrease final tumor volume compared with saline. Mice treated with Doxorubicin plus Zoledronic acid have statistically significantly smaller final tumor volumes than those treated with Doxorubicin alone[6]. 上海金畔生物科技有限公司 has not independently confirmed the accuracy of these methods. They are for reference only.
|
||||||||||||||||||||||||
| Clinical Trial |
|
||||||||||||||||||||||||
| 分子量 |
579.98 |
||||||||||||||||||||||||
| Formula |
C27H30ClNO11 |
||||||||||||||||||||||||
| CAS 号 |
25316-40-9 |
||||||||||||||||||||||||
| 中文名称 |
盐酸阿霉素;盐酸多柔比星 |
||||||||||||||||||||||||
| 运输条件 |
Room temperature in continental US; may vary elsewhere. |
||||||||||||||||||||||||
| 储存方式 |
4°C, sealed storage, away from moisture and light *In solvent : -80°C, 6 months; -20°C, 1 month (sealed storage, away from moisture and light) |
||||||||||||||||||||||||
| 溶解性数据 |
In Vitro:
DMSO : 35.71 mg/mL (61.57 mM; ultrasonic and warming and heat to 60°C) H2O : 20 mg/mL (34.48 mM; Need ultrasonic) 配制储备液
*
请根据产品在不同溶剂中的溶解度选择合适的溶剂配制储备液;一旦配成溶液,请分装保存,避免反复冻融造成的产品失效。 In Vivo:
请根据您的实验动物和给药方式选择适当的溶解方案。以下溶解方案都请先按照 In Vitro 方式配制澄清的储备液,再依次添加助溶剂: ——为保证实验结果的可靠性,澄清的储备液可以根据储存条件,适当保存;体内实验的工作液,建议您现用现配,当天使用; 以下溶剂前显示的百
|
||||||||||||||||||||||||
| 参考文献 |
|
所有产品仅用作科学研究或药证申报,我们不为任何个人用途提供产品和服务